
Water: It’s Not Just H20
By Karen Dallimore
Features Housing ResearchIt comes carrying its own baggage
Water. It comes carrying its own baggage of inorganic and organic
material, then we make it all more complicated by adding sanitizers,
acidifiers, medications, nutritional supplements and vaccines to the
mix.
Water. It comes carrying its own baggage of inorganic and organic material, then we make it all more complicated by adding sanitizers, acidifiers, medications, nutritional supplements and vaccines to the mix.
This prompts the question: What kind of chemical soup are we creating?
Canadian Poultry Magazine asked Dr. Susan Watkins, Associate Professor at the University of Arkansas and Poultry Extension Specialist addressing production line issues, to explain some of the chemical interactions that may be going on behind the lines in our poultry barn water supply.
Dr. Watkins and her team of researchers have been studying the interactions of some common water additives and updating our knowledge about their effects on our modern birds. “There was a lot of work done 20 years ago into water quality, but we’re dealing with a different bird now.” She explained that our modern broiler or turkey is now much more efficient but the price we pay is that they are much more prone to stress, and sub- optimal water can be stressful for the birds.
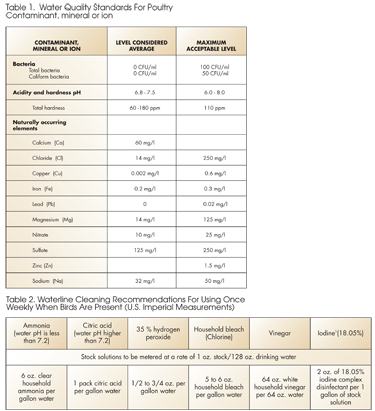 Chemical Actions and Interactions
Chemical Actions and Interactions
While some chemical interactions can be synergistic, actually helping one another to work better, creating the wrong chemical interaction can lead to reduced effectiveness of the additives, not to mention the potential health consequences to both birds and humans. The following are some of the actions and interactions Dr. Watkins says that we know for certain.
Hydrogen peroxide and chlorine are not compatible and should not be run at the same time. Both are oxidizers and they will turn on each other.
Copper sulfate, an antimicrobial and antifungal agent, actually enhances the effectiveness of hydrogen peroxide so they can be run at the same time with separate medicators, however copper sulfate is not compatible with chlorine.
Organic iodine (not the inorganic ‘brown jug stuff’ that is only a sanitizer) is very compatible with chlorine. This combination can help to prevent a full-blown respiratory infection if it is used early enough to treat snicking, a loose croupy cough. Watkins recommends a stock solution of 6 oz. of organic iodine in a gallon of water then 1 oz. of this to a gallon of drinking water (U.S. Imperial measurement), and running this in a separate medicator while you are running your chlorine and acidifier with other injectors.
Dual injection of chlorine and ammonia will create chloramines, another type of sanitizer that can be a good alternative to bleach, particularly if bleach has been used extensively in a system. Watkins cautions: do not mix the chlorine and ammonia or any acid product in the same stock solution – this can create chlorine gas that can be deadly!
Chlorine is not compatible with anything that acts as a reducing agent, which would include most mineral water additives. For example, products that contain copper, sulfur (not bound sulfur), or iron will tie up the chlorine, leaving it unavailable to work effectively as a sanitizer. The effectiveness of the minerals is not likely being affected, but that would be a subject of further research.
Iodine, like chlorine, works better at a lower pH. The recommended range for both is between 4 and 7. Another sanitizer, chlorine dioxide, is effective from a pH of 3 to 9, but it also works best in the lower pH range.
Antibiotics and pH
When it comes to dispensing antibiotics through the water system, the results will vary depending on the antibiotic you’re using. A pH above 7 works best when you are using penicillin and sulfa drugs, so Watkins recommends turning off the water acidifiers and adding ammonia with a second injector. Adding these antibiotics to water with a low pH will actually turn the resulting mixture into an insoluble product.
Chlortetracycline and tetracycline work best in a low pH range. If you have high alkaline water, adding citric acid actually enhances absorption of these products.
Vaccines and pH
Some growers will distribute vaccines via the water system. Vaccines are typically proteins, so any time you run vaccines it should be at a pH above 4 or you will run the risk of denaturing the viral proteins and rendering the vaccine less effective or useless.
Dr. Watkins has been receiving a lot of calls regarding slime blooms in water systems after the use of antibiotics. These fungal growths are catching growers by surprise — literally clogging drinkers — but it makes perfect sense that such growth should occur, particularly with a dirty water system. The antibiotic disrupts the microbial population in the waterlines just as it does in the gut, allowing microbes such as yeast and mold to grow unchecked. She recommends a thorough cleaning to remove the slime, but cautions that using an acidifier such as citric acid will only make it worse.
Effect of Temperature
One other pearl of water wisdom concerns the temperature of the water used to mix your stock solutions. Dr. Watkins suggests that adding dry acidifier products to room temperature water will provide more uniform stock solutions than if cold water is used. All sanitizers lose efficacy at colder temperatures. Chlorine, chlorine dioxide and ozone are all temperature sensitive, with colder water slowing down their reaction times.
Some of the future research at the University of Arkansas will continue to test different combinations of products and evaluate new products as they arrive on the market. Dr. Watkins is also currently working on the possibility of creating an ideal water sanitization program that will help to reduce or prevent the incidence of salmonella in pullet flocks.
Admittedly all of these chemical interactions can make something as simple as water quite complicated. As Dr. Watkins says, one thing she is sure about when it comes to water: it’s almost a daily effort to make sure you’ve got it right.
Print this page